Therapeutic indications1
Cosentyx is indicated for the treatment of moderate to severe plaque psoriasis (PsO) in adults, children and adolescents from the age of 6 years who are candidates for systemic therapy; active psoriatic arthritis (PsA) in adult patients (alone or in combination with methotrexate [MTX]) when the response to previous disease-modifying anti-rheumatic drug therapy has been inadequate; active ankylosing spondylitis (AS) in adults who have responded inadequately to conventional therapy; active non-radiographic axial spondyloarthritis (nr-axSpA) with objective signs of inflammation as indicated by elevated C-reactive protein and/or magnetic resonance imaging evidence in adults who have responded inadequately to non-steroidal anti-inflammatory drugs; active moderate to severe hidradenitis suppurativa (HS; acne inversa) in adults with an inadequate response to conventional systemic HS therapy; active enthesitis-related arthritis (ERA) in patients 6 years and older (alone or in combination with MTX) whose disease has responded inadequately to, or who cannot tolerate, conventional therapy; active juvenile psoriatic arthritis (JPsA) in patients 6 years and older (alone or in combination with MTX) whose disease has responded inadequately to, or who cannot tolerate, conventional therapy.1
*In a meta-analysis of observational studies comprising patients with PsA (n=32,973), compared with the general population.2
†ULTIMATE: Observed data in biologic-naïve patients with PsA receiving Cosentyx 300 mg or 150 mg who were originally randomly assigned to Cosentyx (n=83).8
‡FUTURE 2: Post-hoc analysis of observed data over 104 weeks for the Cosentyx 300 mg treatment group of biologic-naïve patients with this symptom at baseline, including those originally randomly assigned to Cosentyx and placebo-switchers (number of patients originally randomised to Cosentyx 300 mg, n=67; number of evaluable patients at Week 104, n=57).3
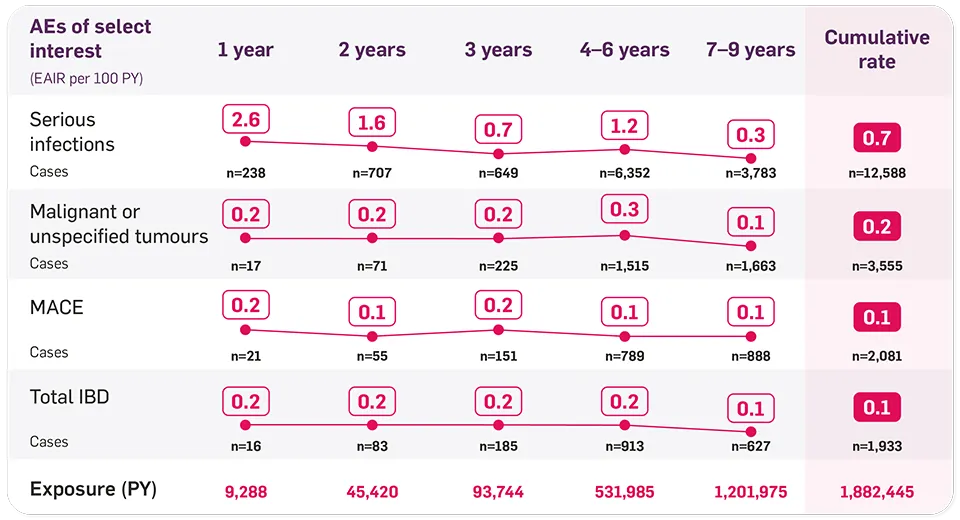
ACR, American College of Rheumatology; AE, adverse event; AS, ankylosing spondylitis; CI, confidence interval; CV, cardiovascular; EAIR, exposure-adjusted incidence rate; ERA, enthesitis-related arthritis; HS, hidradenitis suppurativa; IBD, inflammatory bowel disease; JPsA, juvenile psoriatic arthritis; MACE, major adverse cardiovascular event; MTX, methotrexate; nr-axSpA, non-radiographic axial spondyloarthritis; OR, odds ratio; PsA, psoriatic arthritis; PsO, plaque psoriasis; PSUR, periodic safety update report; PY, patient-year; SmPC, summary of product characteristics; TNFi, tumour necrosis factor inhibitor; VAS, visual analogue scale.
References
Cosentyx® (secukinumab) Summary of Product Characteristics.
Polachek A, et al. Arthritis Care Res 2017;69(1):67–74.
McInnes IB, et al. Arthritis Res Ther 2018;20(1):113.
Novartis Data on File. CAIN457F2342 (FUTURE 5): 2-year HAQ-DI biologic-naive data. February 2021.
Mease PJ, et al. Ann Rheum Did 2018;77:890–897.
Gossec L, et al. Ann Rheum Dis 2017;76(Suppl 2):949.
McInnes IB, et al. Lancet 2015;386(9999):1137–1146.
D'Agostino MA, et al. Semin Arthritis Rheum 2023;63:152259.
Novartis Data on File. Secukinumab (SEC020). April 2025.
Gottlieb AB, et al. Acta Derm Venereol 2022;102:adv00698.
Novartis Data on File. Secukinumab (SEC018). February 2025.
European Medicines Agency. Summary of positive opinion. EMA/CHMP/670627/2015. Available at: https://www.ema.europa.eu/en/documents/smop/chmp-post-authorisation-summary-positive-opinion-cosentyx_en.pdf [Accessed January 2026].