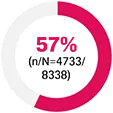
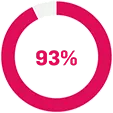
FUTURE 2: was a multicentre, randomised, double-blind, placebo-controlled Phase III trial that evaluated 397 adult patients with active PsA (≥3 swollen and ≥3 tender joints) despite use of NSAIDs, corticosteroids, or DMARDs. Patients received Cosentyx 75 mg (n=99) [Cosentyx 75 mg is not a licensed dose within this population], 150 mg (n=100), 300 mg (n=100), or placebo SC (n=98) at Weeks 0, 1, 2, 3, and 4, followed by the same dose every 4 weeks. Patients who received placebo were re-randomised to receive Cosentyx 150 mg or 300 mg every 4 weeks at Week 16 or Week 24 based on responder status. The primary endpoint was the percentage of patients with ACR20 response at Week 24 — ACR20 results at Week 24: 54% in 300 mg; 15% in placebo; p<0.0001 for placebo vs all doses. At baseline, approximately 65% of patients were biologic-naïve and 47% of patients were treated with concomitant methotrexate.19
AS, ankylosing spondylitis; BSR, British Society for Rheumatology; CVD, cardiovascular disease; ERA, enthesitis-related arthritis; EULAR, European Alliance of Associations for Rheumatology; GRAPPA, Group for Research and Assessment of Psoriasis and Psoriatic Arthritis; HS, hidradenitis suppurativa; IBD, inflammatory bowel disease; IR, inadequate response; JPsA, juvenile psoriatic arthritis; MTX, methotrexate; NAPPA-QOL, nail assessment in psoriasis and psoriatic arthritis quality of life; NNT, number needed to treat; nr-axSpA, non-radiographic axial spondyloarthritis; PsA, psoriatic arthritis; PsO, plaque psoriasis; QoL, quality of life; SC, subcutaneous; SmPC, summary of product characteristics; TNFi, tumour necrosis factor inhibitor; UV, ultraviolet.
References
Cosentyx® (secukinumab) Summary of Product Characteristics.
Ogdie A, et al. J Rheumatol 2021;48(5):698–706.
Tiwari V, et al. In: StatPearls. Psoriatic Arthritis. StatPearls Publishing; 2024. Available at: https://www.ncbi.nlm.nih.gov/books/NBK547710/ [Accessed June 2025].
Ritchlin, CT et al. N Engl J Med 2017;376(10):957–970.
Lee S, et al. P T 2010;35(12):680–689.
Gottlieb AB, et al. J Dermatolog Treat 2006;17(5):279–287.
Polachek A, et al. Arthritis Care Res 2017;69(1):67–74.
Shah K, et al. RMD Open 2017;3(2):e000588.
Armstrong A, et al. J EurAcad Dermatol Venereol 2018;32(12):2200–2207.
Wang TS, Tsai TF. Am J Clin Dermatol 2017;18(1):17-43.
Wilson FC et al. Arthritis Rheum 2009;61(2):233–239.
Vlam K, et al. Rheumatol Ther 2018;5(2):423–436.
Radtke MA, et al. J Dtsch Dermatol Ges 2013;11(3):203–220.
Nail Assessment in Psoriasis and Psoriatic Arthritis. Modular condition-specific questionnaire. Available at: https://nappa-online.com/wp-content/uploads/nappa_short_information.pdf [Accessed June 2025].
Haroon M, et al. Ann Rheum Dis 2015;74(6):1045–1050.
Mease PJ, et al. Drugs 2014;74(4):423–441.
James L, et al. Ther Adv Musculoskelet Dis 2024;16:1–23.
Glintborg B et al. Arthritis Rheum 2013;65(5):1213–1223.
McInnes IB, et al. Lancet 2015;386(9999):1137–1146
Tucker L et al. Rheumatology (Oxford) 2022;61(9):e255–e266.
Gossec L et al. Ann Rheum Dis 2024;83(6):706–719.
Coates LC et al. Net Rev Rheumatol 2022;18(8):465–479.