Explore RLT services
Our all-encompassing service aims to simplify the RLT journey and minimise time-to-treatment for your eligible patients.
This page is intended for UK healthcare professionals and other relevant decision makers only. If you are a member of the public, please click here.
This portal is funded and owned by Novartis Pharmaceuticals UK Ltd and includes content approved by Novartis.
Adverse events reporting information can be found in the footer of this page.
Prescribing information (external link)
LUTATHERA® (lutetium [177Lu] oxodotreotide) is indicated for the treatment of unresectable or metastatic, progressive, well-differentiated (G1 and G2), somatostatin receptor (SSTR)-positive gastroenteropancreatic neuroendocrine tumours (GEP-NETs) in adults.1
LUTATHERA® Summary of Product Characteristics (SmPC) can be found here
Based on pre-clinical data.
With a high affinity for SSTR2, LUTATHERA® is designed to deliver beta radiation directly to tumour cells, disrupting them from within1
The beta radiation is mostly contained within the tumour, limiting damage to neighbouring healthy cells1
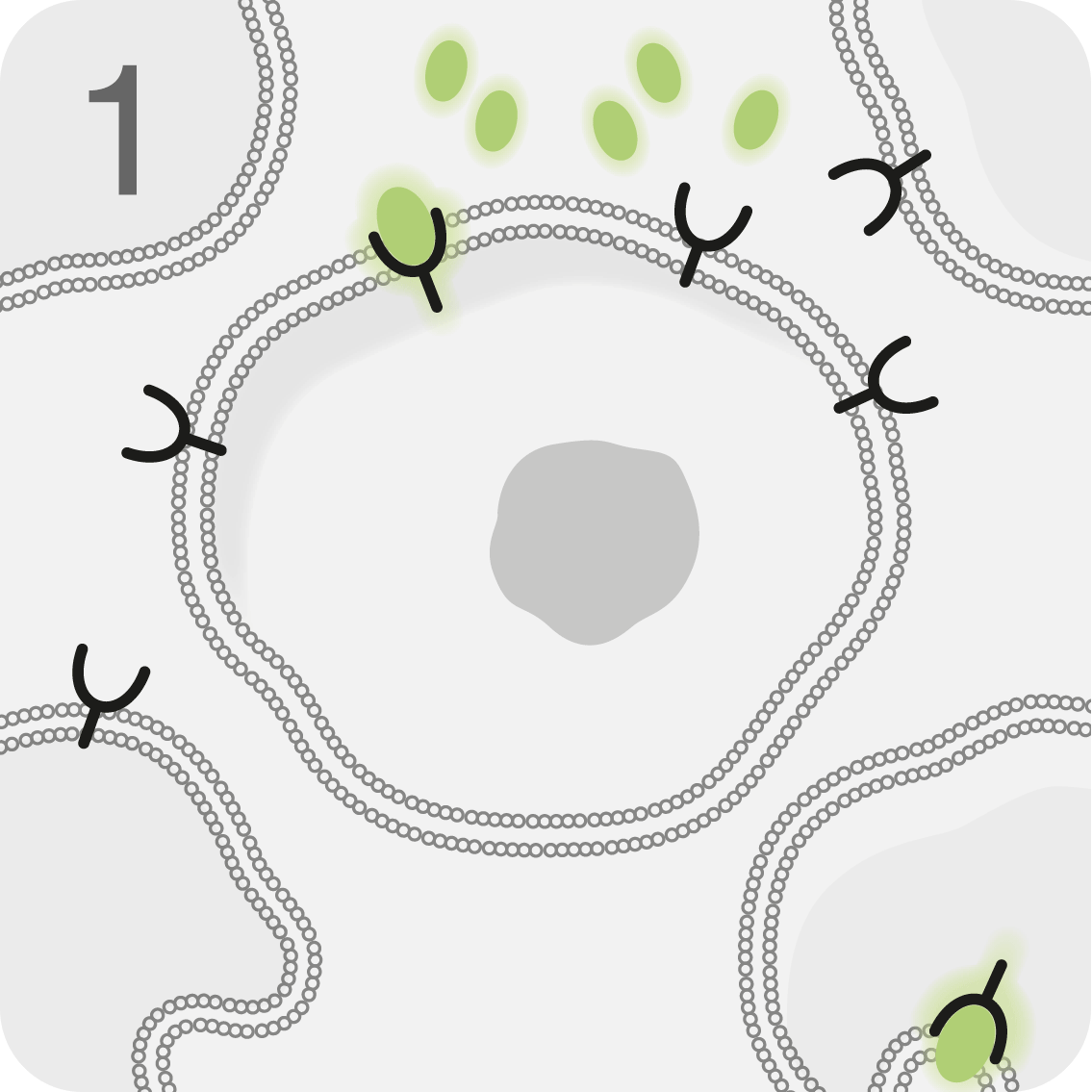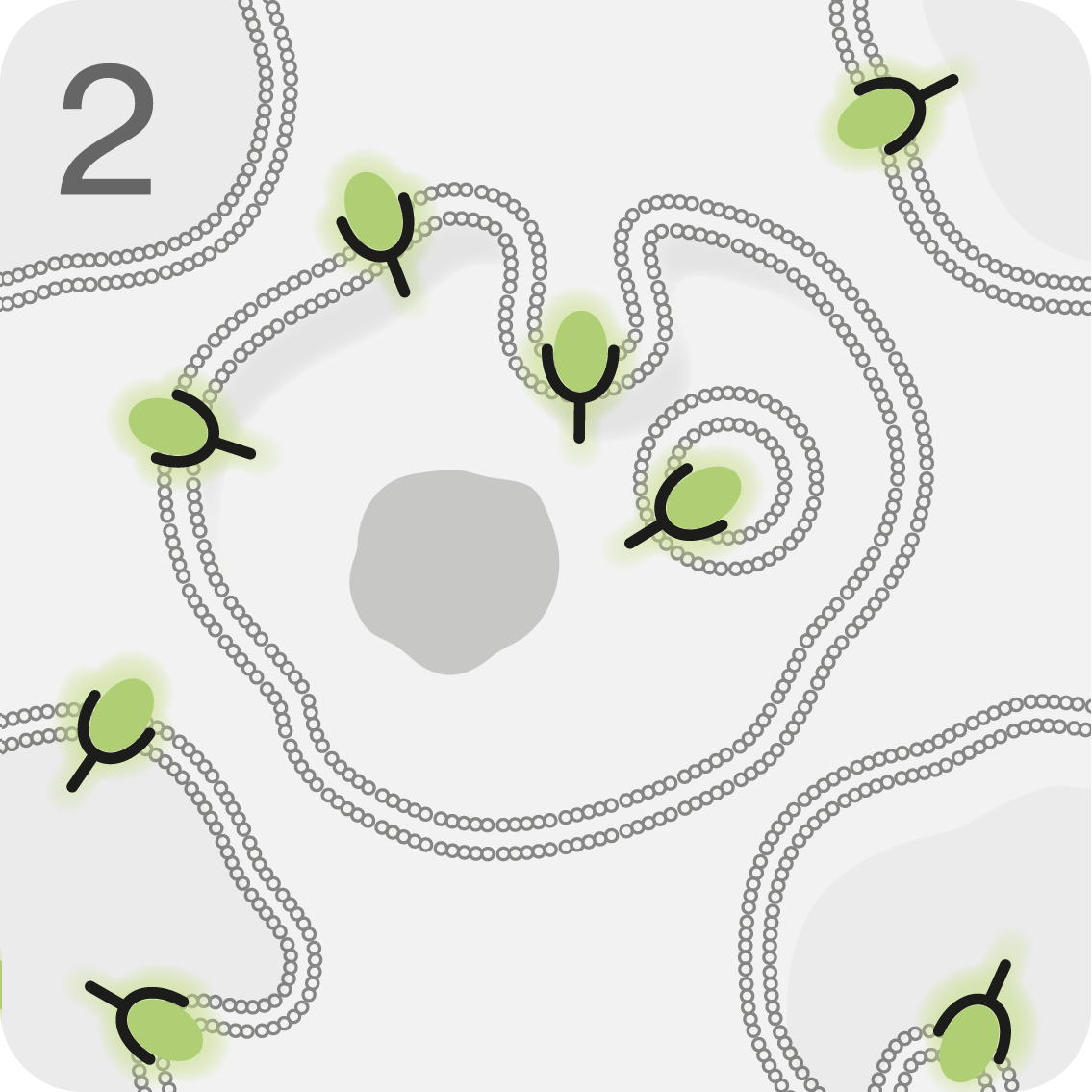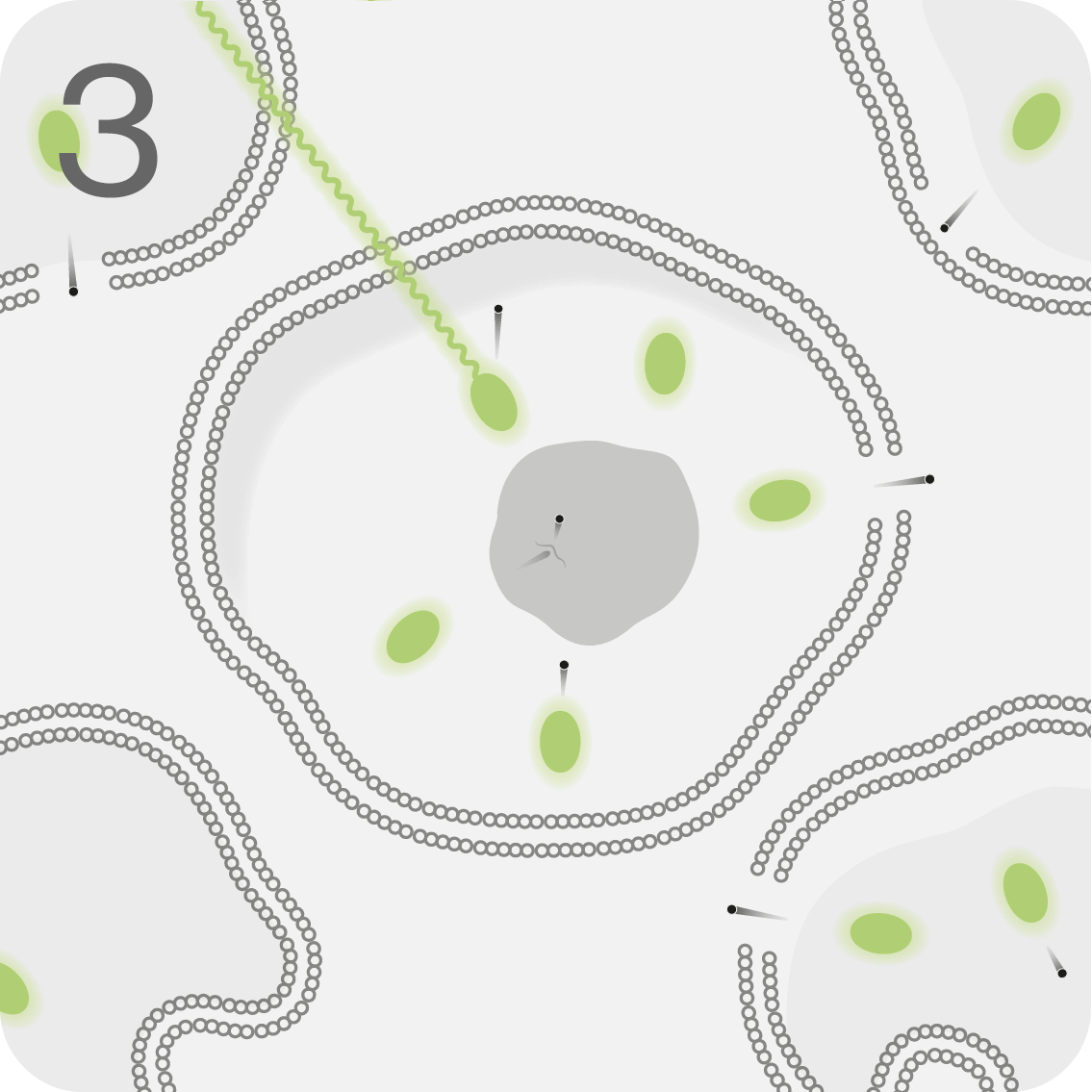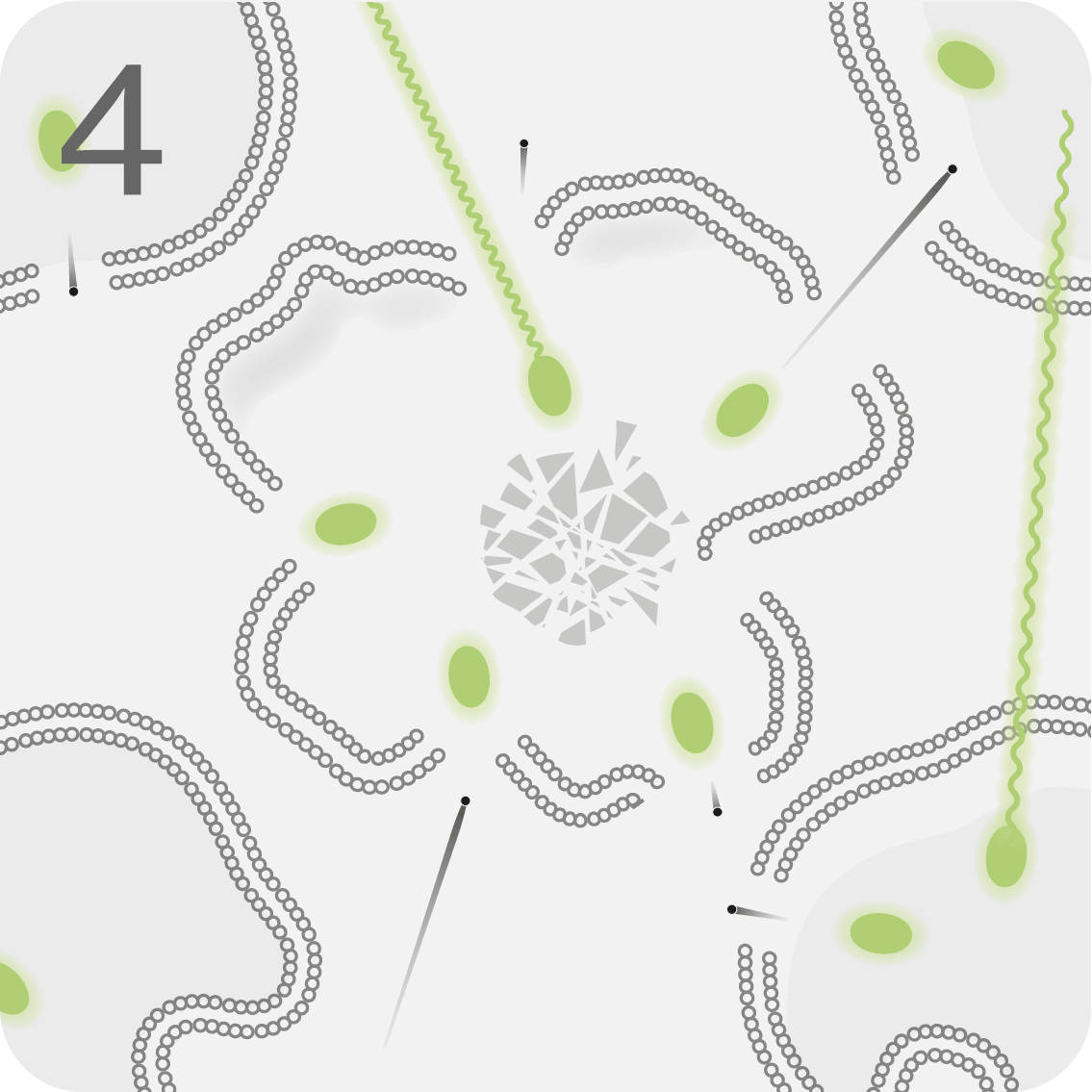
LUTATHERA® is infused into the bloodstream, where it targets and binds to malignant cells overexpressing SSTR2.1
LUTATHERA® is then internalised into the cell.3
In the cell, it delivers beta radiation, with an average tissue penetration of 0.67mm.1
The radiation disrupts the targeted tumour cell from within, with a limited effect on neighbouring normal cells.1
GEP-NET, gastroenteropancreatic neuroendocrine tumour; RLT, radioligand therapy; SmPC, summary of product characteristics; SSTR, somatostatin receptor.
References
LUTATHERA® Summary of Product Characteristics.
Thundimadathil J. J Amino Acids 2012;2012:967347.
Hennrich U & Kopka K. Pharmaceuticals (Basel) 2019;12(3):114.
UK | December 2025 | FA-11462531-1
Adverse events should be reported. Reporting forms and information can be found at www.mhra.gov.uk/yellowcard. Adverse events should also be reported to Novartis online through the pharmacovigilance intake (PVI) tool at www.novartis.com/report, or alternatively email [email protected] or call 01276 698370.
![Hero banner. LUTATHERA® (lutetium [177Lu] oxodotreotide) logo. Hero banner. LUTATHERA® (lutetium [177Lu] oxodotreotide) logo.](/uk-en/sites/pro_novartis_com_uk/files/styles/twoup_layout_desktop_1080/public/2025-08/lutathera-home-hero-banner-hcp-2700_0.png.webp?itok=KiWg7nHk)
![Hero banner. LUTATHERA® (lutetium [177Lu] oxodotreotide) logo. Hero banner. LUTATHERA® (lutetium [177Lu] oxodotreotide) logo.](/uk-en/sites/pro_novartis_com_uk/files/styles/twoup_layout_desktop_1080/public/2025-08/lutathera-home-hero-banner-hcp-1000_0.png.webp?itok=cL3k8yXL)