COSENTYX® (secukinumab) prescribing information (external link)
XOLAIR® (omalizumab) prescribing information (external link)
This page includes promotional content created and funded by Novartis Pharmaceuticals UK Ltd. Novartis products may be mentioned.
Dermatology
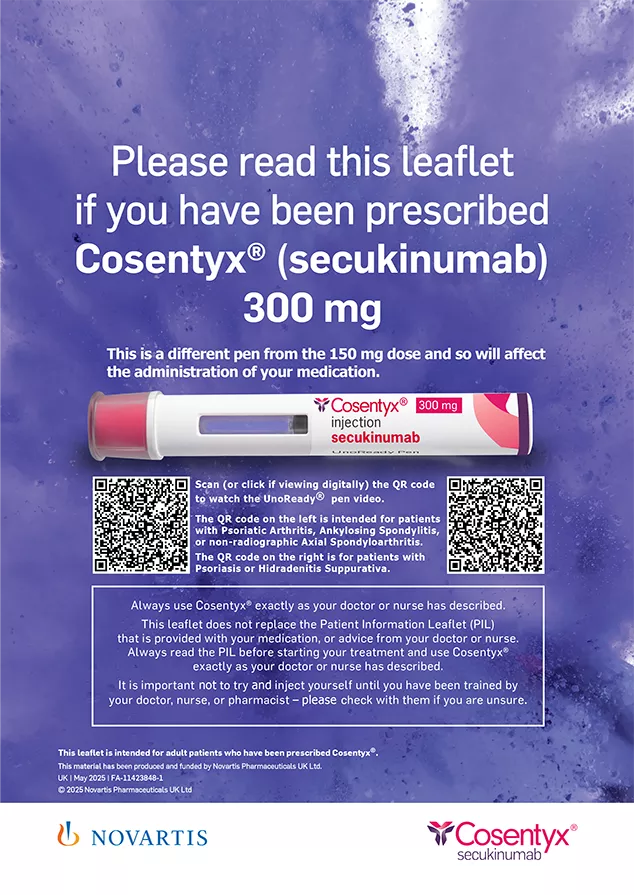
COSENTYX® (secukinumab)
XOLAIR® (omalizumab)
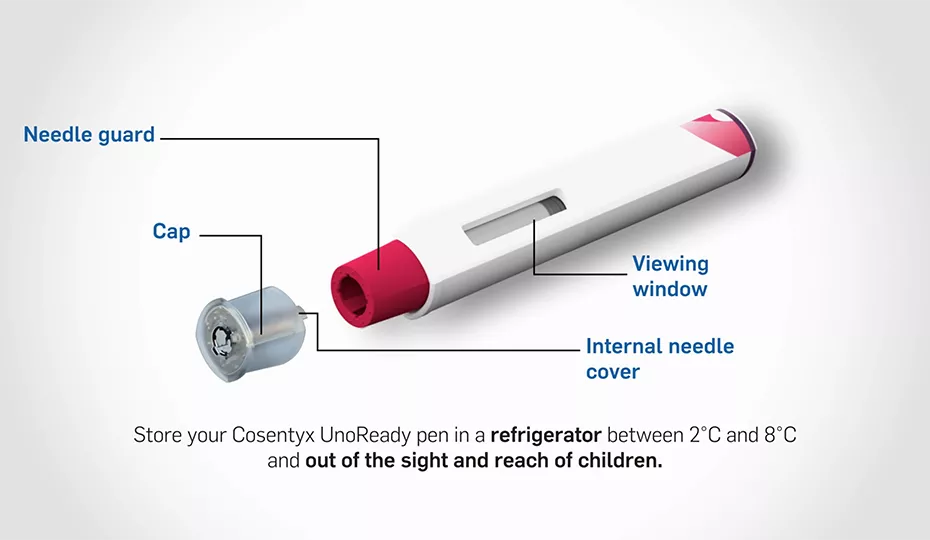
Self-administration starter kit (1): The essentials
Self-administration starter kit (1): The essentials
Self-administration starter kit (2): A guide to self-injection
Self-administration starter kit (2): A guide to self-injection
Self-administration starter kit (3): Things for you to fill in
Self-administration starter kit (3): Things for you to fill in
Self-administration starter kit (4): Important information
Self-administration starter kit (4): Important information
Adverse events should be reported. Reporting forms and information can be found at www.mhra.gov.uk/yellowcard. Adverse events should also be reported to Novartis online through the pharmacovigilance intake (PVI) tool at www.novartis.com/report, or alternatively email [email protected] or call 01276 698370.