Explore RLT services
Our all-encompassing service aims to simplify the RLT journey and minimise time-to-treatment for your eligible patients.
This page is intended for UK healthcare professionals and other relevant decision makers only. If you are a member of the public, please click here.
This portal is funded and owned by Novartis Pharmaceuticals UK Ltd and includes content approved by Novartis.
Adverse events reporting information can be found in the footer of this page.
Pluvicto▼(lutetium [177Lu] vipivotide tetraxetan) prescribing information (external link)
Locametz▼(gozetotide) prescribing information (external link)
Pluvicto®▼ (lutetium [177Lu] vipivotide tetraxetan) is indicated for the treatment of adult patients with:1
Prostate-specific membrane antigen (PSMA)-positive metastatic castration-resistant prostate cancer (mCRPC) who have progressed on or after treatment with androgen receptor pathway inhibitor (ARPI) therapy and are considered appropriate to delay taxane-based chemotherapy
PSMA-positive mCRPC who have been treated with androgen receptor (AR) pathway inhibition and taxane-based chemotherapy or who are not medically suitable for taxanes
Pluvicto Summary of Product Characteristics (SmPC) can be found here
Pluvicto is available in the United Kingdom to eligible private patients.
Locametz®▼ (gozetotide) is for diagnostic use only. Locametz, after radiolabelling with gallium-68, is a radioactive diagnostic agent indicated for the identification of prostate-specific membrane antigen (PSMA)-positive lesions by positron emission tomography (PET) in adult patients with prostate cancer.2
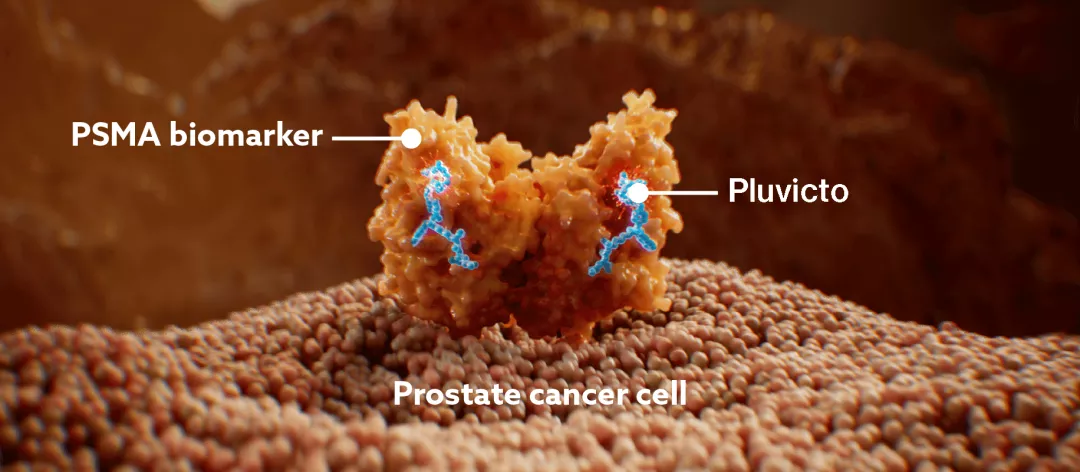
Pluvicto is a PSMA-targeted radioligand therapy (RLT) that delivers DNA-damaging radiation to PSMA-positive bone, nodal, and visceral metastases.1,3–7
Pluvicto targets PSMA-positive cells, including prostate cancer cells.1
Pluvicto comprises 2 key components:
lutetium-177, a cytotoxic radionuclide, and PSMA-617, a PSMA-targeting ligand.1
As determined by pre-clinical research, Pluvicto binds with high affinity to PSMA, a transmembrane protein overexpressed on prostate cancer cells.1
After binding to PSMA, Pluvicto undergoes endocytosis and is internalised into the cell.1,3
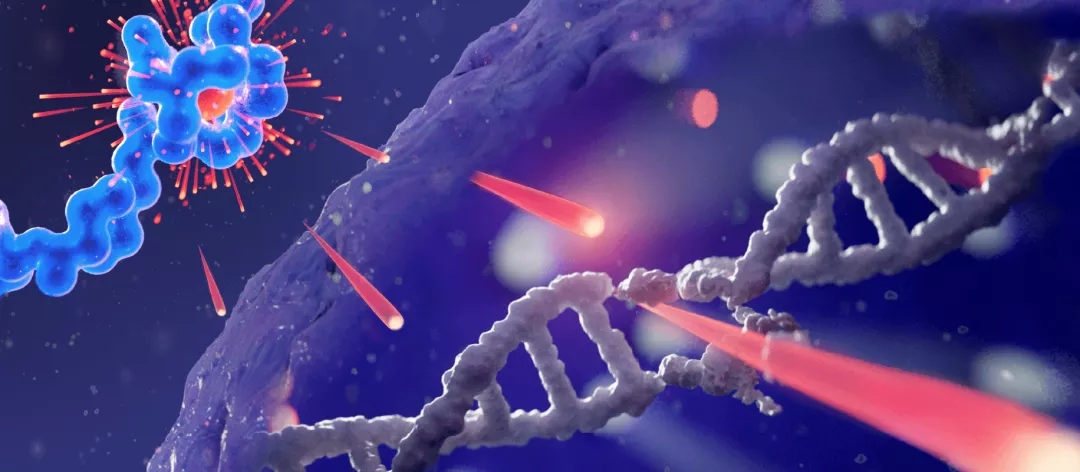
As determined by preclinical research, lutetium-177, (the cytotoxic radionuclide of Pluvicto), emits DNA-damaging radiation within the cell.1,3,5
The short path length of the radiation emitted by Pluvicto (approx. 2 mm max.), causes single- and double-stranded DNA breaks in targeted cells as well as surrounding cells, which can lead to cell death.4,5,10
The safety profile of Pluvicto was evaluated in two trials, PSMAfore and VISION. In PSMAfore, the most common (≥20%) adverse drug reactions (ADRs) occurring in patients who received Pluvicto include: dry mouth (60.8%), fatigue (52.9%), nausea (31.7%), anaemia (27.3%), constipation (22.0%), and decreased appetite (21.6%). The most common grade 3 to 4 ADRs (≥5%) occurring in patients who received Pluvicto include: anaemia (6.2%).1
In VISION, the most common (≥20%) ADRs occurring at a higher incidence in patients who received Pluvicto + BSoC compared to BSoC alone include: fatigue (48.0%), dry mouth (39.3%), nausea (35.7%), anaemia (31.9%), decreased appetite (21.4%) and constipation (20.2%). The most common grade 3 to 4 ADRs (≥5%) occurring at a higher incidence in patients who received Pluvicto + BSoC compared to BSoC alone include: anaemia (12.9%), thrombocytopenia (7.9%), lymphopenia (7.8%) and fatigue (6.6%).1
The data from the PSMAfore and VISION trials cannot be extrapolated to any other lutetium PSMA therapy.
Please refer to the Pluvicto SmPC for full safety information.
177Lu, lutetium-177; ADR, adverse drug reaction; AR, androgen receptor; ARPI, androgen receptor pathway inhibitor; BSoC, best standard of care; DNA, deoxyribonucleic acid; mCRPC, metastatic castration-resistant prostate cancer; MoA, mechanism of action; PET, positron emission tomography; PSMA, prostate-specific membrane antigen; RLT, radioligand therapy; SmPC, summary of product characteristics.
References
Pluvicto®▼ (lutetium [177Lu] vipivotide tetraxetan) Summary of Product Characteristics.
Locametz®▼ (gozetotide) Summary of Product Characteristics.
Benešová M, et al. J Nucl Med 2015;56(6):914–920.
Fendler WP, et al. J Nucl Med 2017;58(11):1786–1792.
Hofman MS, et al. Lancet Oncol 2018;19(6):825–833.
Sartor O, et al. N Engl J Med 2021;385(12):1091–1103.
Violet J, et al. J Nucl Med 2019;60(4):517–523.
Liu H, et al. Cancer Res 1998;58(18):4055–4060.
Rajasekaran SA, et al. Mol Biol Cell 2003;14(12):4835–4845.
Ruigrok EAM, et al. Eur J Nucl Med Mol Imaging 2021;48(5):1339–1350.
Maffey-Steffan J, et al. Eur J Nucl Med Mol Imaging 2020;47(3):695–712.
Vlachostergios PJ, et al. Front Oncol 2021;11:630589.
Woythal N, et al. J Nucl Med 2018;59(2):238–243.
Schmuck S, et al. J Nucl Med 2017;58(12):1962–1968.
UK | February 2026 | FA-11470709-2
Adverse events should be reported. Reporting forms and information can be found at www.mhra.gov.uk/yellowcard. Adverse events should also be reported to Novartis online through the pharmacovigilance intake (PVI) tool at www.novartis.com/report, or alternatively email [email protected] or call 01276 698370.